Summary
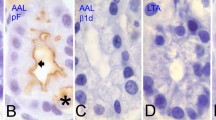
Antibodies specific for chromogranin A, B or C have been used to detect immunohistochemically these three anionic proteins. Pancreatic A, B and PP cells, gut argentaffin EC, argyrophil ECL and gastrin G cells, thyroid C cells, parathyroid cells, adrenal medullary cells, pituitary TSH, FSH and LH cells as well as some axons of visceral nerves have been found to react with chromogranin A antibodies. Pancreatic A, gut EC and G, adrenal medullary and pituitary cells as well as some gut nerve fibers showed chromogranin B immunoreactivity. Chromogranin C immunoreactivity has been detected in pancreatic A, pyloric D1, intestinal L, thyroid C, adrenal medullary and pituitary cells, as well as in some gut neurons and nerve fibers. No crossreactivity has been found in immunohistochemical tests between chromogranins A, B or C and costored monoamines or peptide hormones/prohormones, from which chromogranins can be separated by selective extraction during fixation. On both morphological and chemical grounds a relationship seems to exist between chromogranin A and Grimelius' argyrophilia. Sialooligosaccharide chains of chromogranin A and, possibly, chromogranins' phosphoserine/phosphothreonine groups, seem to interact with guanidyl, amino, and/or imidazole groups of non-chromogranin components to form silver complexing sites accounting for granules' argyrophilia, which can be removed or blocked without affecting chromogranin immunoreactivities. The abundant anionic groups of the three proteins should contribute substantially to granules' basophilia, the partly “masked” pattern of which supports the existence of a close interaction of such groups with other components of secretory granules, including monoamines and peptide hormones or prohormones. Chromogranins could play a role in hormone postranslational biosynthesis and intragranular packaging.
Similar content being viewed by others
References
Bodian D (1937) The staining of paraffin sections of nervous tissue with activated protargol. The role of fixatives. Anat Rec 69:152–162
Buffa R, Solovieva I, Fiocca R, Giorgino S, Rindi G, Solcia E, Mochizuchi T, Yanaihara C, Yanaihara N (1982) Localization of bombesin and GRP (Gastrin Releasing Peptide) sequences in gut nerves or endocrine cells. Histochemistry 76:457–467
Cohn DV, Zangerle R, Fischer-Colbrie R, Chu LLH, Elting JJ, Hamilton JW, Winkler H (1982) Similarity of secretory protein I from parathyroid gland to chromogranin A from adrenalin medulla. Proc Natl Acad Sci USA 79:6056–6059
Dockerty K, Steiner DF (1982) Post-translational proteolysis in polypeptide hormone biosynthesis. Annu Rev Physiol 44:625–638
Falkensammer G, Fischer-Colbrie R, Richter K, Winkler H (1985) Cell-free and cellular synthesis of chromogranin A and B of bovine adrenal medulla. Neuroscience 14:735–746
Fiocca R, Sessa F, Tenti P, Usellini L, Capella C, O'Have MMT, Solcia E (1983) Pancreatic polypeptide (PP) cells in the PP-rich lobe of the human pancreas are identified ultrastructurally and immunocytochemically as F cells. Histochemistry 77:511–523
Fischer-Colbrie R, Frischenschlager I (1985) Immunologic characterization of secretory proteins of chromaffin granules: chromogranins A, chromogranins B, and enkephalin-containing peptides. J Neurochem 44:1854–1861
Fischer-Colbrie R, Schachinger M, Zangerle R, Winkler H (1982) Dopamine B-hydroxylase and other glycoproteins from the soluble content and the membranes of adrenal chromaffin granules: isolation and carbohydrate analysis. J Neurochem 38:725–732
Fischer-Colbrie R, Lassmann H, Hagn C, Winkler H (1985) Immunological studies of the distribution of chromogranin A and B in endocrine and nervous tissues. Neuroscience 16: 547–555
Fischer-Colbrie R, Hagn C, Kilpatrick L, Winkler H (1986) Chromogranin C: a third component of the acidic proteins in chromaffin granules. J Neurochem (In press)
Grimelius L (1968) A silver nitrate stain for α2 cells in human pancreatic islets. Acta Soc Med Upsal 73:243–270
Gurd FRN, Wilcox PhE (1956) Complex formation between metallic cations and proteins, peptides, and amino acids. Adv. Prot Chem 11:311–427
Hougaard DM, Larsson L-I (1982) Polyamine cytochemistry. Use of a novel o-phtalaldehyde method for visualizing spermidine and spermine. Comparisons to the formaldehyde fluorescamine method. Histochemistry 76:247–259
Hsu S-M, Rainel L, Fanger H (1981) Use of avidin-biotin-peroxidase complex (ABC) in immunoperoxidase techniques. J Histochem Cytochem 29:577–580
Kiang W-L, Krusius T, Finne J, Margolis RU, Margolis RK (1982) Glycoproteins and proteoglycans of the chromaffin granule matrix. J Biol Chem 257:1651–1659
Lischwe MA, Smetana K, Olson MOJ, Busch H (1979) Proteins C23 and B23 are the major nucleolar silver staining proteins. Life Sci 25:701–708
Lloyd RV, Wilson BS (1983) Specific endocrine tissue marker defined by a monoclonal antibody. Science 222:628–630
Lloyd RV, Mervak T, Schmidt K, Khazaeli MB, Wilson BS (1984) Immunohistochemical detection of chromogranins, neuron-specific enolase, and HCG in gastroenteropancreatic neuroendocrine tumors. Lab Invest 50:35/A
O'Connor DT (1983) Chromogranin: widespread immunoreactivity in polypeptide hormone producing tissues and in serum. Regul Peptides 6:263–280
O'Connor DT, Burton D, Deftos LJ (1983) Cromogranin A: immunohistology reveals its universal occurrence in normal polypeptide hormone producing endocrine glands. Life Sci 33:1657–1664
Petkov P, Galabova R (1969) Zinkverteilung im Pankreas einiger Säuger. Acta Histochem 37:93–109
Polak JM, Van Noorden S (1983) Immunocytochemistry. Wright-PSG, Bristol
Rosa P, Zanini A (1983) Purification of a sulfated secretory protein from the adenohypophysis. Immunochemical evidence that similar macromolecules are present in other glands. Eur J Cell Biol 31:94–98
Rosa P, Fumagalli G, Zanini A, Huttner WB (1985a) The major tyrosine-sulfated protein of the bovine anterior pituitary is a secretory protein present in gonadotrophs, thyrotrophs, mammotrophs, and corticotrophs. J Cell Biol 100:928–937
Rosa P, Hille A, Lee RWH, Zanini A, De Camilli P (1985b) Secretogranins I and II. Two tyrosine-sulfated secretory proteins common to a variety of cells secreting peptides by the regulated pathway. J Cell Biol 101:1999–2011
Satoh K, Busch H (1981) Silver staining of phosphoserine and phosphothreonine in nucleolar and other phosphoproteins. Cell Biol Int Rep 5:857–866
Schäfer H-J, Klöppel G (1974) The significance of calcium in insulin secretion. Ultrastructural studies of identification and localization of calcium in activated and inactivated B cells of mice. Virch Arch A Pathol Histol 362:231–245
Solcia E, Vassallo G, Capella C (1968) Selective staining of endocrine cells by basic dyes after acid hydrolysis. Stain Technol 43:257–263
Solcia E, Capella C, Vassallo G (1969a) Lead-haematoxylin as a stain for endocrine cells. Significance of staining and comparison with other selective methods. Histochemie 20:116–126
Solcia E, Sampietro R, Capella C (1969b) Differential staining of catecholamines, 5-hydroxytryptamine and related compounds in aldehyde-fixed tissues. Histochemie 17:273–283
Solcia E, Capella C, Buffa R, Frigerio B (1976) Histochemical and ultrastructural studies on the argentaffin an argyrophil cells of the gut. In: Coupland RE, Fujita T (eds) Chromaffin, enterochromaffin and related cells. Elsevier, Amsterdam, pp 209–225
Solcia E, Capella C, Buffa R, Tenti C, Rindi G, Cornaggia M (1986) Antigenic markers of neuroendocrine tumors: their diagnostic and prognostic value. In: Fenoglio CM, Weinsten RS (eds) New concepts in neoplasia as applied to diagnostic pathology. Internat Acad Pathol, Vol 27. Williams and Wilkins, Baltimore
Somogyi P, Hodgson AJ, De Potter RW, Fischer-Colbrie R, Schober M, Winkler H, Chubb W (1984) Chromogranin immunoreactivity in the central nervous system. Immunochemical characterization, distribution and relationship to catecholamine and enkephalin pathways. Brain Res Rev 8:193–230
Stefanini M, De Martino C, Zamboni L (1967) Fixation of ejaculated spermatozoa for electron microscopy. Nature 216:173–174
Takatsuki K, Schneider AB, Shin KY, Sherwood LM (1981) Extraction, purification, and partial characterization of bovine parathyroid secretory protein. J Biol Chem 256:2342–2345
Varndell IM, Bishop AE, Lloyd RV, Wilson BS, Grimelius L, Pearse AGE, Polak JM (1984) Chromogranin A and Grimelius' argyrophilia. A possible correlation. Dig Dis Sci 29:Suppl 935
Wilson BS, Lloyd RV (1984) Detection of chromogranin in neuroendocrine cells with a monoclonal antibody. Am J Pathol 115:458–468
Winkler H (1976) The composition of adrenal chromaffin granules: an assessment of controversial results. Neuroscience 1:65–80
Winkler H, Westhead E (1980) The molecular organization of adrenal chromaffin granules. Neuroscience 5:1803–1823
Winkler H, Falkensammer G, Patzak A, Fischer-Colbrie R, Schober M, Weber A (1984) Life cycle of the catecholaminergic vesicle: from biogenesis to secretion. In: Vizi ES, Magyar K (eds) Regulation of transmitter function. Proc 5th Meeting Eur Soc Neurochem, pp 65–73
Winkler H, Apps DK, Fischer-Colbrie R (1986) The molecular function of adrenal chromaffin granules: established facts and controversial results. Neuroscience (In press)
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Rindi, G., Buffa, R., Sessa, F. et al. Chromogranin A, B and C immunoreactivities of mammalian endocrine cells. Histochemistry 85, 19–28 (1986). https://doi.org/10.1007/BF00508649
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00508649