Key Points
-
The important role of stromal cells in the regulation of immune responses is becoming increasingly appreciated. In addition to providing a supportive lattice for lymphocytes by expressing adhesion molecules, the stroma directs lymphocyte migration by secreting chemokines and can interact with lymphocytes through cytokine secretion.
-
In addition to the relevance of stromal cells in the initiation of immune responses, there is evidence that these cells regulate the generation and maintenance of immunological memory. Follicular dendritic cells are involved in the generation of memory B cells and plasma cells during germinal centre reactions.
-
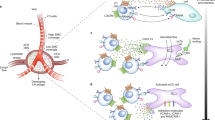
Bone marrow stroma is thought to have a role in the maintenance of several types of memory lymphocytes. It is composed of mesenchymal stromal cells, endothelial cells, osteoblasts and adipocytes.
-
Long-lived plasma cells have been shown to dock on a subpopulation of CXCL12+VCAM1+ mesenchymal stromal cells that is thought to form specific survival niches for those cells, thus regulating the size of the memory plasma cell compartment. Resting memory CD4+ T cells also reside in the bone marrow but, in contrast to plasma cells, they are found in close association with IL-7+VCAM1+ stromal cells.
-
Memory B cells are thought to be localized in the spleen, but their microanatomical niche has not been defined in detail. The localization of resting memory CD8+ T cells is currently unclear.
-
A deeper understanding of stromal cell populations and subpopulations in different anatomical locations is necessary to identify the molecular mechanisms involved in the maintenance of immunological memory.
Abstract
Immunological memory is a hallmark of the adaptive immune system. Plasma cells and memory B and T cells collectively provide protective immunity and effective secondary immune responses to invading pathogens. Here, we discuss how mesenchymal stromal cells regulate immunological memory by organizing defined numbers of dedicated survival niches for plasma cells and memory T cells in the bone marrow and also, to a lesser extent, in secondary lymphoid organs. An understanding of the biology of mesenchymal stromal cells and their interaction with cells of the immune system is key to fully understanding immunological memory.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Friedenstein, A. J., Chailakhyan, R. K., Latsinik, N. V., Panasyuk, A. F. & Keiliss-Borok, I. V. Stromal cells responsible for transferring the microenvironment of the hemopoietic tissues. Cloning in vitro and retransplantation in vivo. Transplantation 17, 331–340 (1974).
Mueller, S. N. & Germain, R. N. Stromal cell contributions to the homeostasis and functionality of the immune system. Nature Rev. Immunol. 9, 618–629 (2009).
Ahmed, R. & Gray, D. Immunological memory and protective immunity: understanding their relation. Science 272, 54–60 (1996).
Zinkernagel, R. M. et al. On immunological memory. Annu. Rev. Immunol. 14, 333–367 (1996).
Radbruch, A. et al. Competence and competition: the challenge of becoming a long-lived plasma cell. Nature Rev. Immunol. 6, 741–750 (2006).
Kalia, V., Sarkar, S., Gourley, T. S., Rouse, B. T. & Ahmed, R. Differentiation of memory B and T cells. Curr. Opin. Immunol. 18, 255–264 (2006).
Nakayama, T. & Yamashita, M. Initiation and maintenance of Th2 cell identity. Curr. Opin. Immunol. 20, 265–271 (2008).
Zhou, L., Chong, M. M. & Littman, D. R. Plasticity of CD4+ T cell lineage differentiation. Immunity 30, 646–655 (2009).
Lohning, M. et al. Long-lived virus-reactive memory T cells generated from purified cytokine-secreting T helper type 1 and type 2 effectors. J. Exp. Med. 205, 53–61 (2008).
Radbruch, A. & Rajewsky, K. in The Molecular Biology of B cells (eds Alt, F. W., Honjo, T. & Neuberger M.) 247–259 (Elsevier Academic, London, 2004).
Nossal, G. J. Genetic control of lymphopoiesis, plasma cell formation, and antibody production. Int. Rev. Exp. Pathol. 1, 1–72 (1962).
Miller, J. J. An autoradiographic study of plasma cell and lymphocyte survival in rat popliteal lymph nodes. J. Immunol. 92, 673–681 (1964).
Holt, P. G., Sedgwick, J. D., O'Leary, C., Krska, K. & Leivers, S. Long-lived IgE- and IgG-secreting cells in rodents manifesting persistent antibody responses. Cell. Immunol. 89, 281–289 (1984).
Ho, F., Lortan, J. E., MacLennan, I. C. & Khan, M. Distinct short-lived and long-lived antibody-producing cell populations. Eur. J. Immunol. 16, 1297–1301 (1986).
Manz, R. A., Thiel, A. & Radbruch, A. Lifetime of plasma cells in the bone marrow. Nature 388, 133–134 (1997). This paper shows that plasma cells are long lived and reside in the bone marrow.
Slifka, M. K., Antia, R., Whitmire, J. K. & Ahmed, R. Humoral immunity due to long-lived plasma cells. Immunity 8, 363–372 (1998).
Manz, R. A., Lohning, M., Cassese, G., Thiel, A. & Radbruch, A. Survival of long-lived plasma cells is independent of antigen. Int. Immunol. 10, 1703–1711 (1998).
Hoyer, B. F. et al. Short-lived plasmablasts and long-lived plasma cells contribute to chronic humoral autoimmunity in NZB/W mice. J. Exp. Med. 199, 1577–1584 (2004).
Benner, R., Meima, F., van der Meulen, G. M. & van Muiswinkel, W. B. Antibody formation in mouse bone marrow. I. Evidence for the development of plaque-forming cells in situ. Immunology 26, 247–255 (1974).
Tokoyoda, K., Egawa, T., Sugiyama, T., Choi, B. I. & Nagasawa, T. Cellular niches controlling B lymphocyte behavior within bone marrow during development. Immunity 20, 707–718 (2004). This study shows that in the bone marrow CXCL12-expressing mesenchymal stromal cells interact with pre-pro-B cells and plasma cells, and IL-7-expressing mesenchymal stromal cells interact with pro-B cells.
Tokoyoda, K. et al. Professional memory CD4+ T lymphocytes preferentially reside and rest in the bone marrow. Immunity 30, 721–730 (2009). This study shows that memory CD4+ T cells are maintained on IL-7-expressing mesenchymal stromal cells.
Tokoyoda, K., Zehentmeier, S., Chang, H. D. & Radbruch, A. Organization and maintenance of immunological memory by stroma niches. Eur. J. Immunol. 39, 2095–2099 (2009).
Greenberger, J. S. The hematopoietic microenvironment. Crit. Rev. Oncol. Hematol. 11, 65–84 (1991).
Cattoretti, G., Schiro, R., Orazi, A., Soligo, D. & Colombo, M. P. Bone marrow stroma in humans: anti-nerve growth factor receptor antibodies selectively stain reticular cells in vivo and in vitro. Blood 81, 1726–1738 (1993).
Clark, B. R. & Keating, A. Biology of bone marrow stroma. Ann. NY Acad. Sci. 770, 70–78 (1995).
Krebsbach, P. H., Kuznetsov, S. A., Bianco, P. & Robey, P. G. Bone marrow stromal cells: characterization and clinical application. Crit. Rev. Oral Biol. Med. 10, 165–181 (1999).
Bianco, P., Riminucci, M., Gronthos, S. & Robey, P. G. Bone marrow stromal stem cells: nature, biology, and potential applications. Stem Cells 19, 180–192 (2001).
Morikawa, S. et al. Prospective identification, isolation, and systemic transplantation of multipotent mesenchymal stem cells in murine bone marrow. J. Exp. Med. 206, 2483–2496 (2009).
Funk, P. E., Stephan, R. P. & Witte, P. L. Vascular cell adhesion molecule 1-positive reticular cells express interleukin-7 and stem cell factor in the bone marrow. Blood 86, 2661–2671 (1995).
Honczarenko, M. et al. Human bone marrow stromal cells express a distinct set of biologically functional chemokine receptors. Stem Cells 24, 1030–1041 (2006).
Pittenger, M. F. et al. Multilineage potential of adult human mesenchymal stem cells. Science 284, 143–147 (1999).
Bentley, S. A., Alabaster, O. & Foidart, J. M. Collagen heterogeneity in normal human bone marrow. Br. J. Haematol. 48, 287–291 (1981).
Weiss, L. & Chen, L. T. The organization of hematopoietic cords and vascular sinuses in bone marrow. Blood Cells 1, 617–638 (1975).
Patt, H. M. & Maloney, M. A. Bone marrow regeneration after local injury: a review. Exp. Hematol. 3, 135–148 (1975).
Chan, P. Y. & Aruffo, A. VLA-4 integrin mediates lymphocyte migration on the inducible endothelial cell ligand VCAM-1 and the extracellular matrix ligand fibronectin. J. Biol. Chem. 268, 24655–24664 (1993).
Springer, T. A. Traffic signals on endothelium for lymphocyte recirculation and leukocyte emigration. Annu. Rev. Physiol. 57, 827–872 (1995).
Jacobsen, K., Kravitz, J., Kincade, P. W. & Osmond, D. G. Adhesion receptors on bone marrow stromal cells: in vivo expression of vascular cell adhesion molecule-1 by reticular cells and sinusoidal endothelium in normal and γ-irradiated mice. Blood 87, 73–82 (1996).
Newman, P. J. et al. PECAM-1 (CD31) cloning and relation to adhesion molecules of the immunoglobulin gene superfamily. Science 247, 1219–1222 (1990).
Clark, B. R., Gallagher, J. T. & Dexter, T. M. Cell adhesion in the stromal regulation of haemopoiesis. Baillieres Clin. Haematol. 5, 619–652 (1992).
Zuckerman, K. S. & Wicha, M. S. Extracellular matrix production by the adherent cells of long-term murine bone marrow cultures. Blood 61, 540–547 (1983).
Nilsson, S. K. et al. Immunofluorescence characterization of key extracellular matrix proteins in murine bone marrow in situ. J. Histochem. Cytochem. 46, 371–377 (1998).
Keating, A. et al. Donor origin of the in vitro haematopoietic microenvironment after marrow transplantation in man. Nature 298, 280–283 (1982).
Cooper, A. R. & MacQueen, H. A. Subunits of laminin are differentially synthesized in mouse eggs and early embryos. Dev. Biol. 96, 467–471 (1983).
Waller, E. K., Huang, S. & Terstappen, L. Changes in the growth properties of CD34+, CD38− bone marrow progenitors during human fetal development. Blood 86, 710–718 (1995).
Simmons, P. J. & Torok-Storb, B. Identification of stromal cell precursors in human bone marrow by a novel monoclonal antibody, STRO-1. Blood 78, 55–62 (1991).
Keating, A., Whalen, C. K. & Singer, J. W. Cultured marrow stromal cells express common acute lymphoblastic leukaemia antigen (CALLA): implications for marrow transplantation. Br. J. Haematol. 55, 623–628 (1983).
Dennis, J. E. & Charbord, P. Origin and differentiation of human and murine stroma. Stem Cells 20, 205–214 (2002).
Mythreye, K. & Blobe, G. C. Proteoglycan signaling co-receptors: roles in cell adhesion, migration and invasion. Cell. Signal. 21, 1548–1558 (2009).
Friedenstein, A. J., Petrakova, K. V., Kurolesova, A. I. & Frolova, G. P. Heterotopic of bone marrow. Analysis of precursor cells for osteogenic and hematopoietic tissues. Transplantation 6, 230–247 (1968).
Caplan, A. I. Mesenchymal stem cells. J. Orthop. Res. 9, 641–650 (1991).
Horwitz, E. M. et al. Clarification of the nomenclature for MSC: The International Society for Cellular Therapy position statement. Cytotherapy 7, 393–395 (2005).
Dominici, M. et al. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 8, 315–317 (2006).
Uccelli, A., Moretta, L. & Pistoia, V. Mesenchymal stem cells in health and disease. Nature Rev. Immunol. 8, 726–736 (2008).
Bartholomew, A. et al. Mesenchymal stem cells suppress lymphocyte proliferation in vitro and prolong skin graft survival in vivo. Exp. Hematol. 30, 42–48 (2002).
Di Nicola, M. et al. Human bone marrow stromal cells suppress T-lymphocyte proliferation induced by cellular or nonspecific mitogenic stimuli. Blood 99, 3838–3843 (2002).
Krampera, M. et al. Bone marrow mesenchymal stem cells inhibit the response of naive and memory antigen-specific T cells to their cognate peptide. Blood 101, 3722–3729 (2003).
Meisel, R. et al. Human bone marrow stromal cells inhibit allogeneic T-cell responses by indoleamine 2, 3-dioxygenase-mediated tryptophan degradation. Blood 103, 4619–4621 (2004).
Glennie, S., Soeiro, I., Dyson, P. J., Lam, E. W. & Dazzi, F. Bone marrow mesenchymal stem cells induce division arrest anergy of activated T cells. Blood 105, 2821–2827 (2005).
Plumas, J. et al. Mesenchymal stem cells induce apoptosis of activated T cells. Leukemia 19, 1597–1604 (2005).
Aggarwal, S. & Pittenger, M. F. Human mesenchymal stem cells modulate allogeneic immune cell responses. Blood 105, 1815–1822 (2005).
Pabst, R. Plasticity and heterogeneity of lymphoid organs. What are the criteria to call a lymphoid organ primary, secondary or tertiary? Immunol. Lett. 112, 1–8 (2007).
Sipkins, D. A. et al. In vivo imaging of specialized bone marrow endothelial microdomains for tumour engraftment. Nature 435, 969–973 (2005).
Holmes, K., Roberts, O. L., Thomas, A. M. & Cross, M. J. Vascular endothelial growth factor receptor-2: structure, function, intracellular signalling and therapeutic inhibition. Cell. Signal. 19, 2003–2012 (2007).
Kopp, H. G., Hooper, A. T., Avecilla, S. T. & Rafii, S. Functional heterogeneity of the bone marrow vascular niche. Ann. NY Acad. Sci. 1176, 47–54 (2009).
Kiel, M. J., Yilmaz, O. H., Iwashita, T., Terhorst, C. & Morrison, S. J. SLAM family receptors distinguish hematopoietic stem and progenitor cells and reveal endothelial niches for stem cells. Cell 121, 1109–1121 (2005).
Sapoznikov, A. et al. Perivascular clusters of dendritic cells provide critical survival signals to B cells in bone marrow niches. Nature Immunol. 9, 388–395 (2008).
Pereira, J. P., An, J., Xu, Y., Huang, Y. & Cyster, J. G. Cannabinoid receptor 2 mediates the retention of immature B cells in bone marrow sinusoids. Nature Immunol. 10, 403–411 (2009).
Maeda, K., Kosco-Vilbois, M. H., Burton, G. F., Szakal, A. K. & Tew, J. G. Expression of the intercellular adhesion molecule-1 on high endothelial venules and on non-lymphoid antigen handling cells: interdigitating cells, antigen transporting cells and follicular dendritic cells. Cell Tissue Res. 279, 47–54 (1995).
Szabo, M. C., Butcher, E. C. & McEvoy, L. M. Specialization of mucosal follicular dendritic cells revealed by mucosal addressin-cell adhesion molecule-1 display. J. Immunol. 158, 5584–5588 (1997).
Balogh, P., Aydar, Y., Tew, J. G. & Szakal, A. K. Appearance and phenotype of murine follicular dendritic cells expressing VCAM-1. Anat. Rec. 268, 160–168 (2002).
Allen, C. D. & Cyster, J. G. Follicular dendritic cell networks of primary follicles and germinal centers: phenotype and function. Semin. Immunol. 20, 14–25 (2008).
Qin, D. et al. Fc gamma receptor IIB on follicular dendritic cells regulates the B cell recall response. J. Immunol. 164, 6268–6275 (2000).
Cyster, J. G. et al. Follicular stromal cells and lymphocyte homing to follicles. Immunol. Rev. 176, 181–193 (2000).
Kapasi, Z. F. et al. Follicular dendritic cell (FDC) precursors in primary lymphoid tissues. J. Immunol. 160, 1078–1084 (1998).
Munoz-Fernandez, R. et al. Follicular dendritic cells are related to bone marrow stromal cell progenitors and to myofibroblasts. J. Immunol. 177, 280–289 (2006).
Bachmann, M. F., Odermatt, B., Hengartner, H. & Zinkernagel, R. M. Induction of long-lived germinal centers associated with persisting antigen after viral infection. J. Exp. Med. 183, 2259–2269 (1996).
Kundig, T. M. et al. On T cell memory: arguments for antigen dependence. Immunol. Rev. 150, 63–90 (1996).
Katakai, T. et al. A novel reticular stromal structure in lymph node cortex: an immuno-platform for interactions among dendritic cells, T cells and B cells. Int. Immunol. 16, 1133–1142 (2004).
Sixt, M. et al. The conduit system transports soluble antigens from the afferent lymph to resident dendritic cells in the T cell area of the lymph node. Immunity 22, 19–29 (2005).
Luther, S. A., Tang, H. L., Hyman, P. L., Farr, A. G. & Cyster, J. G. Coexpression of the chemokines ELC and SLC by T zone stromal cells and deletion of the ELC gene in the plt/plt mouse. Proc. Natl Acad. Sci. USA. 97, 12694–12699 (2000).
Okada, T. & Cyster, J. G. CC chemokine receptor 7 contributes to Gi-dependent T cell motility in the lymph node. J. Immunol. 178, 2973–2978 (2007).
Link, A. et al. Fibroblastic reticular cells in lymph nodes regulate the homeostasis of naive T cells. Nature Immunol. 8, 1255–1265 (2007). This paper shows IL-7-expressing stromal cells in SLOs.
Forster, R., Davalos-Misslitz, A. C. & Rot, A. CCR7 and its ligands: balancing immunity and tolerance. Nature Rev. Immunol. 8, 362–371 (2008).
Gretz, J. E., Kaldjian, E. P., Anderson, A. O. & Shaw, S. Sophisticated strategies for information encounter in the lymph node: the reticular network as a conduit of soluble information and a highway for cell traffic. J. Immunol. 157, 495–499 (1996).
Balogh, P., Aydar, Y., Tew, J. G. & Szakal, A. K. Appearance and phenotype of murine follicular dendritic cells expressing VCAM-1. Anat. Rec. 268, 160–168 (2002).
Roozendaal, R., Mebius, R. E. & Kraal, G. The conduit system of the lymph node. Int. Immunol. 20, 1483–1487 (2008).
McCuskey, R. S. Morphological mechanisms for regulating blood flow through hepatic sinusoids. Liver 20, 3–7 (2000).
MacLennan, I. C. Germinal centers. Annu. Rev. Immunol. 12, 117–139 (1994).
Fliedner, T. M. et al. Bone marrow structure and its possible significance for hematopoietic cell renewal. Ann. NY Acad. Sci. 459, 73–84 (1985).
Kondrack, R. M. et al. Interleukin 7 regulates the survival and generation of memory CD4 cells. J. Exp. Med. 198, 1797–1806 (2003).
Li, J., Huston, G. & Swain, S. L. IL-7 promotes the transition of CD4 effectors to persistent memory cells. J. Exp. Med. 198, 1807–1815 (2003).
McKinstry, K. K. et al. Rapid default transition of CD4 T cell effectors to functional memory cells. J. Exp. Med. 204, 2199–2211 (2007).
Surh, C. D. & Sprent, J. Homeostasis of naive and memory T cells. Immunity 29, 848–862 (2008).
Hargreaves, D. C. et al. A coordinated change in chemokine responsiveness guides plasma cell movements. J. Exp. Med. 194, 45–56 (2001).
Hauser, A. E. et al. Chemotactic responsiveness toward ligands for CXCR3 and CXCR4 is regulated on plasma blasts during the time course of a memory immune response. J. Immunol. 169, 1277–1282 (2002).
Mamani-Matsuda, M. et al. The human spleen is a major reservoir for long-lived vaccinia virus-specific memory B cells. Blood 111, 4653–4659 (2008). This study shows that memory B cells reside in the spleen.
Martinez-Gamboa, L. et al. Role of the spleen in peripheral memory B-cell homeostasis in patients with autoimmune thrombocytopenia purpura. Clin. Immunol. 130, 199–212 (2009).
Dogan, I. et al. Multiple layers of B cell memory with different effector functions. Nature Immunol. 10, 1292–1299 (2009).
Nossal, G. J., Szenberg, A., Ada, G. L. & Austin, C. M. Single cell studies on 19s antibody production. J. Exp. Med. 119, 485–502 (1964).
Cassese, G. et al. Inflamed kidneys of NZB / W mice are a major site for the homeostasis of plasma cells. Eur. J. Immunol. 31, 2726–2732 (2001).
Benson, M. J. et al. The dependence of plasma cells and independence of memory B cells on BAFF and APRIL. J. Immunol. 180, 3655–3659 (2008).
Cassese, G. et al. Plasma cell survival is mediated by synergistic effects of cytokines and adhesion-dependent signals. J. Immunol. 171, 1684–1690 (2003).
Seddon, B., Legname, G., Tomlinson, P. & Zamoyska, R. Long-term survival but impaired homeostatic proliferation of naive T cells in the absence of p56lck. Science 290, 127–131 (2000).
Boyman, O., Letourneau, S., Krieg, C. & Sprent, J. Homeostatic proliferation and survival of naive and memory T cells. Eur. J. Immunol. 39, 2088–2094 (2009).
Seddon, B., Tomlinson, P. & Zamoyska, R. Interleukin 7 and T cell receptor signals regulate homeostasis of CD4 memory cells. Nature Immunol. 4, 680–686 (2003).
Hataye, J., Moon, J. J., Khoruts, A., Reilly, C. & Jenkins, M. K. Naive and memory CD4+ T cell survival controlled by clonal abundance. Science 312, 114–116 (2006). This paper shows that the pool size of memory CD4+ T H cells is regulated by clonal abundance and suggests the existence of specific survival niches in the body.
Tomayko, M. M. et al. Systematic comparison of gene expression between murine memory and naive B cells demonstrates that memory B cells have unique signaling capabilities. J. Immunol. 181, 27–38 (2008).
Becker, T. C., Coley, S. M., Wherry, E. J. & Ahmed, R. Bone marrow is a preferred site for homeostatic proliferation of memory CD8 T cells. J. Immunol. 174, 1269–1273 (2005).
Mazo, I. B. et al. Bone marrow is a major reservoir and site of recruitment for central memory CD8+ T cells. Immunity 22, 259–270 (2005). References 108 and 109 shows that memory CD8+ T cells preferentially reside in the bone marrow.
MacLennan, I. C. et al. Extrafollicular antibody responses. Immunol. Rev. 194, 8–18 (2003).
Haaijman, J. J., Schuit, H. R. & Hijmans, W. Immunoglobulin-containing cells in different lymphoid organs of the CBA mouse during its life-span. Immunology 32, 427–434 (1977).
Terstappen, L. W., Johnsen, S., Segers-Nolten, I. M. & Loken, M. R. Identification and characterization of plasma cells in normal human bone marrow by high-resolution flow cytometry. Blood 76, 1739–1747 (1990).
Suematsu, S. et al. IgG1 plasmacytosis in interleukin 6 transgenic mice. Proc. Natl Acad. Sci. USA. 86, 7547–7551 (1989).
Jego, G. et al. Reactive plasmacytoses are expansions of plasmablasts retaining the capacity to differentiate into plasma cells. Blood 94, 701–712 (1999).
Sykes, M. & Nikolic, B. Treatment of severe autoimmune disease by stem-cell transplantation. Nature 435, 620–627 (2005).
Alexander, T. et al. Depletion of autoreactive immunologic memory followed by autologous hematopoietic stem cell transplantation in patients with refractory SLE induces long-term remission through de novo generation of a juvenile and tolerant immune system. Blood 113, 214–223 (2009).
Podojil, J. R., Turley, D. M. & Miller, S. D. Therapeutic blockade of T-cell antigen receptor signal transduction and costimulation in autoimmune disease. Adv. Exp. Med. Biol. 640, 234–251 (2008).
Niesner, U. et al. Autoregulation of Th1-mediated inflammation by twist1. J. Exp. Med. 205, 1889–1901 (2008).
Acknowledgements
We thank S. Zehentmeier, A. Hanazawa, H.-D. Chang, T. Doerner, A. Hegazy, F. Hiepe and M. Löhning for valuable discussions. This work was supported by grants: SFB 421, SFB 633 and SFB 650 of the German Research Foundation (DFG). K.T. was a research fellow of the Alexander von Humboldt foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
Related links
FURTHER INFORMATION
Deutsches Rheuma-Forschungzentrum (DRFZ), A Leibniz Institute homepage
Glossary
- Stromal cells
-
Cells of non-lymphoid origin that form the framework of each organ. These cells can support adhesion, proliferation and survival of distinct cell subsets.
- Central tolerance
-
Tolerance to self that is created at the level of the central lymphoid organs. Developing T cells (in the thymus), and B cells (in the bone marrow) that strongly recognize self antigen face deletion or marked suppression.
- Follicular dendritic cell
-
A cell type that is normally found only in the germinal centres of lymphoid tissue. It presents antigen to selected B cells and provides survival signals required for affinity maturation.
- Memory B cell
-
An antigen-experienced B cell that expresses high-affinity antibodies and quickly differentiates into a plasma cell in antigen recall responses.
- Plasma cell
-
A terminally differentiated quiescent B cell that develops from a plasmablast and is characterized by an ability to secrete large amounts of antibody.
- Sinusoid
-
A specialized blood vessel in haematopoietic tissues through which venous circulation occurs. It has thin walls formed by a discontinuous, irregularly shaped endothelium that allows cells to pass in and out of circulation.
- Germinal centre
-
A lymphoid structure that arises within follicles after immunization with, or exposure to, a T cell-dependent antigen. It is specialized for facilitating the development of high-affinity, long-lived plasma cells and memory B cells.
- High endothelial venule
-
A specialized venule that occurs in secondary lymphoid organs, except the spleen. High endothelial venules allow continuous transmigration of lymphocytes as a consequence of the constitutive expression of adhesion molecules and chemokines their luminal surface.
- Plasmablast
-
A dividing antibody-secreting cell of the B cell lineage that has migratory potential. These cells can further mature into plasma cells, which do not divide.
Rights and permissions
About this article
Cite this article
Tokoyoda, K., Hauser, A., Nakayama, T. et al. Organization of immunological memory by bone marrow stroma. Nat Rev Immunol 10, 193–200 (2010). https://doi.org/10.1038/nri2727
Published:
Issue Date:
DOI: https://doi.org/10.1038/nri2727
This article is cited by
-
Bortezomib: a proteasome inhibitor for the treatment of autoimmune diseases
Inflammopharmacology (2021)
-
Quantitative spatial analysis of haematopoiesis-regulating stromal cells in the bone marrow microenvironment by 3D microscopy
Nature Communications (2018)
-
Memory CD4+ T cell subsets in tumor draining lymph nodes of breast cancer patients: A focus on T stem cell memory cells
Cellular Oncology (2018)
-
The chronically inflamed central nervous system provides niches for long-lived plasma cells
Acta Neuropathologica Communications (2017)