Summary
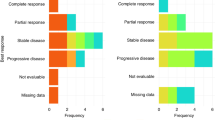
We performed this dose-finding study with a fixed dose of cisplatin and increasing doses of paclitaxel given every 2 weeks to determine the maximum tolerable dose of this schedule. Sixty-four patients with advanced oesophageal cancer were treated with a cisplatin dose of 60 mg m–2 and increasing doses of paclitaxel from 100 mg m–2 up to 200 mg m–2 both administered over 3 h for a maximum of six cycles in patients with stable disease or eight cycles in responding patients. Patients were retreated when the granulocytes were > 0.75 × 109 l–1 and the platelets > 75 × 109 l–1. The dose of paclitaxel could be increased to 200 mg m–2 without encountering dose limiting haematological toxicity. At the dose levels 190 mg m–2 and 200 mg m–2 of paclitaxel cumulative sensory neurotoxicity became the dose-limiting toxicity. The dose intensity of paclitaxel calculated over six cycles rose from 50 mg m–2 per week to 85 mg m–2 per week. Only three episodes of granulocytopenic fever were encountered out of a total of 362 cycles of treatment. Of the 59 patients evaluable for response, 31 (52%) had a partial or complete response. In a biweekly schedule with a fixed dose of 60 mg m–2 cisplatin it is possible to increase the dose of paclitaxel to 180 mg m–2. At higher dose levels, neurotoxicity becomes the dose-limiting toxicity. The observed response rate warrants further investigation of this schedule.
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Ajani, J. A., Ilson, D. H., Daugherty, K., Pazdur, R., Lynch, P. M. & Kelsen, D. P. (1994). Activity of taxol in patients with squamous cell carcinoma and adenocarcinoma of the esophagus. J Natl Cancer Inst 86: 1086–1091.
Ajani, J. A., Ilson, D. H. & Kelsen, D. P. (1996). Paclitaxel in the treatment of patients with upper gastrointestinal carcinomas. Semin Oncol 23: 55–58.
Connelly, E., Markman, M., Kennedy, A., Webster, K., Kulp, B., Peterson, G. & Belinson, J. (1996). Paclitaxel delivered as 3-hr infusion with cisplatin in patients with gynecologic cancers: unexpected incidence of neurotoxicity. Gynecol Oncol 62: 166–168.
Daly, J. M., Karnell, L. & Menck, H. R. (1996). National Cancer Data Base report on esophageal carcinoma. Cancer 78: 1820–1828.
Eisenhauer, E. A., ten Bokkel Huinink, W. W., Swenerton, K. D., Gianni, L., Myles, J., van der Burg, M. E. L., Kerr, I., Vermorken, J. B., Buser, K., Colombo, N., Bacon, M., Santabarbara, P., Onetto, N., Winograd, W. & Canetta, R. (1994). European-Canadian randomized trial of paclitaxel in relapsed ovarian cancer: high-dose versus low-dose and long versus short infusion. J Clin Oncol 12: 2654–2666.
Fennelly, D., Aghajanian, C., Shapiro, F., O’Flaherty, C., McKenzie, M., O’Connor, C., Tong, W., Norton, L. & Spriggs, D. (1997). Phase I and pharmacologic study of paclitaxel administered weekly in patients with relapsed ovarian cancer. J Clin Oncol 15: 187–192.
Gelmon, K. A., O’Reilly, S. E., Tolcher, A. W., Campbell, C., Bryce, C., Ragaz, J., Coppin, C., Plenderleith, I. H., Ayers, D., McDermott, B., Nakashima, L., Healy, D. & Onetto, N. (1996a). Phase I/II trial of bi-weekly paclitaxel and cisplatin in the treatment of metastatic breast cancer. J Clin Oncol 14: 1185–1191.
Gelmon, K. A., Murray, N., Melosky, B., Shah, A., Page, R., Dulude, H. & Rielly, S. (1996b). Phase I/II study of biweekly paclitaxel and cisplatin in non-small-cell lung cancer. Proc Am Soc Clin Oncol 15: 405 (Abstract)
Gordon, A. N., Stringer, C. A., Matthews, C. M., Willis, D. L. & Nemunaitis, J. (1997). Phase I dose escalation of paclitaxel in patients with advanced ovarian cancer receiving cisplatin: rapid development of neurotoxicity is dose-limiting. J Clin Oncol 15: 1965–1973.
Herskovic, A., Martz, K., al-Sarraf, M., Leichman, L., Brindle, J., Vaitkevicius, V., Cooper, J., Byhardt, R., Davis, L. & Emami, B. (1992). Combined chemotherapy and radiotherapy compared with radiotherapy alone in patients with cancer of the esophagus. N Engl J Med 326: 1593–1598.
Hilkens, P. H., van der Burg, M. E., Moll, J. W., Planting, A. S., van Putten, W. L., Vecht, C. J. & van den Bent, M. J. (1995). Neurotoxicity is not enhanced by increased dose intensities of cisplatin administration. Eur J Cancer 31: 678–681.
Holmes, F. A., Walters, R. S., Theriault, R. L., Forman, A. D., Newton, L. K., Raber, M. N., Buzdar, A. U., Frye, D. K. & Hortobagyi, G. N. (1990). Phase II trial of Taxol, an active drug in the treatment of metastatic breast cancer. J Natl Cancer Inst 82: 1247–1249.
Kelsen, D., Ginsberg, R., Qian, C., Gunderson, L., Mortimer, J., Estes, N., Hailer, D., Ajani, J., Kocha, W., Roth, J. & Minsky, B. (1997). Chemotherapy followed by operation versus operation alone in the treatment of patients with localized esophageal cancer: a preliminary report of intergroup study 113 (RTOG 89-11). Proc Am Soc Clin Oncol 16: 276 (Abstract)
Kok, T. C., van der Gaast, A., Stoter, G., Tilanus, H., Eykenboom, W., Dees, J., van Overhagen, H. & Splinter, T. (1996). Cisplatin and etoposide in esophageal cancer. A phase II study. Br J Cancer 74: 980–984.
Kok, T. C. (1997). Chemotherapy in esophageal cancer: a review. Cancer Treat Rev 23: 65–85.
Kok, T. C., van Lanschot, J., Siersema, P. D., van Overhagen, H. & Tilanus, H. W. (1997). Neoadjuvant chemotherapy in operable esophageal squamous cell cancer: final report of a phase III multicenter randomized controlled trial. Proc Am Soc Clin Oncol 16: 277 (Abstract)
McCaskill-Stevens, W., Ansari, R., Fisher, W., Pennington, K., Dobbs, C., Gonin, R., Schaefer, S., Loesch, D. & Sledge, G. (1996). Phase II study of biweekly cisplatin and paclitaxel in the treatment of metastatic breast cancer. Proc Am Soc Clin Oncol 15: 120 (Abstract)
McGuire, W. P., Hoskins, W. J., Brady, M. F., Kucera, P. R., Partridge, E. E., Look, K. Y., Clarke-Pearson, D. L. & Davidson, M. (1996). Cyclophosphamide and cisplatin compared with paclitaxel and cisplatin in patients with stage III and IV ovarian cancer. N Engl J Med 334: 1–6.
O’Reilly, S. & Forastiere, A. (1995). Is surgery necessary with multimodality treatment of esophageal cancer? Ann Oncol 6: 519–521.
Parimoo, D., Garcia, A., Muggia, F., Dimery, I., Rogers, M. & Jeffers, S. (1996). Tolerance of paclitaxel (Taxol) 3-hr infusion with G-CSF on a biweekly schedule. Proc Am Soc Clin Oncol 15: 181 (Abstract)
Roth, J. A. & Putnam, J. B. Jr (1994). Surgery for cancer of the esophagus. Sem Oncol 21: 453–461.
Rowinsky, E. K., Chaudhry, V., Forastiere, A., Sartorius, S. E., Ettinger, D. S., Grochow, L. B., Lubejko, B. G., Cornblath, D. R. & Donehower, R. C. (1993). Phase I and pharmacologic study of paclitaxel and cisplatin with granulocyte colony-stimulating factor: neuromuscular toxicity is dose-limiting. J Clin Oncol 11: 2010–2020.
Sørenson, J. B., Wedervang, K. & Dombernowsky, P. (1997). Preliminary results of a phase II study of paclitaxel and cisplatin in patients with non-small cell lung cancer. Semin Oncol 24: S12.18–S12.20.
Sparano, J. A., Neuberg, D., Glick, J. H., Robert, N. J., Goldstein, L. J., Sledge, G. W. & Wood, W. (1997). Phase II trial of bi-weekly paclitaxel and cisplatin in advanced breast carcinoma: an Eastern Cooperative Oncology Group study. J Clin Oncol 15: 1880–1004.
Swenerton, K., Hoskins, P., Stuart, G., Batist, G., Pike, J., Onetto, N., Fisher, B. & Eisenhauer, E. (1996). A phase I study of biweekly paclitaxel/cisplatin as initial therapy for advanced ovarian cancer. A study of the National Cancer Institute of Canada Clinical Trials Group. Ann Oncol 7: 1077–1079.
Author information
Authors and Affiliations
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Gaast, A., Kok, T., Kerkhofs, L. et al. Phase I study of a biweekly schedule of a fixed dose of cisplatin with increasing doses of paclitaxel in patients with advanced oesophageal cancer. Br J Cancer 80, 1052–1057 (1999). https://doi.org/10.1038/sj.bjc.6690462
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bjc.6690462
Keywords
This article is cited by
-
Phase I-II study of biweekly paclitaxel administration with fixed-dose-rate cisplatin in advanced gastric cancer
Gastric Cancer (2006)
-
Prognostic factors for survival in patients with advanced oesophageal cancer treated with cisplatin-based combination chemotherapy
British Journal of Cancer (2003)
-
Phase II study of bi-weekly administration of paclitaxel and cisplatin in patients with advanced oesophageal cancer
British Journal of Cancer (2002)
-
New developments in the treatment of esophageal cancer
Current Oncology Reports (2002)